Search
- All
- News
- Opinion
- Commentary
- Media Miss
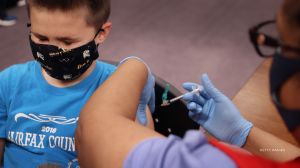
Update (Jan. 5, 2022): An advisory panel to the Centers for Disease Control and Prevention (CDC) voted Wednesday to approve a Food and Drug Administration (FDA) decision regarding the Pfizer COVID-19 vaccine booster in kids. That decision, in part, allowed for kids aged 12-15 to get boosted. The CDC panel also said teens should get the…

For the second time in as many days, the Food and Drug Administration (FDA) has approved a COVID-19 pill. Thursday’s approval was for Merck’s COVID-19 pill, also known as Molnupiravir. This followed the FDA approving the Pfizer COVID-19 pill, also known as Paxlovid, on Wednesday. Unlike the Pfizer pill, which is approved for adults and…

The Food and Drug Administration (FDA) approved the Pfizer COVID-19 pill for emergency use Wednesday. The video above shows clips of the White House COVID-19 Response Team discussing the approval. This marks the first time U.S. health regulators have approved an at-home treatment for COVID-19 since the pandemic began. “This authorization provides a new tool…

The Food and Drug Administration (FDA) announced Thursday American women are no longer required to pick up abortion pills. Under the change, women will be able to get a prescription via an online consultation and receive the pills through the mail. The FDA stopped enforcing the in-person requirement earlier due to the pandemic. Thursday’s decision…
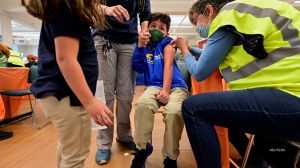
Both the Food and Drug Administration (FDA) and the Centers for Disease Control and Prevention CDC approved Pfizer boosters for kids 16 and 17 years old Thursday, a day after the Senate blocked the Biden administration’s vaccine mandate for businesses employing 100 or more workers. The FDA and CDC moves serve as the first to…

In a narrow Tuesday afternoon vote of 13-10, an advisory panel to the Food and Drug Administration (FDA) approved Merck’s COVID-19 pill. Molnupiravir is an oral antiviral drug initially hailed as a potential game changer in the battle against Covid since it can be taken at home instead of at a hospital like other treatments.…

A day after reports started coming out that the Food and Drug Administration was looking to approve Pfizer’s COVID-19 boosters in kids ages 16 and 17, new details are leaving more questions than answers as to the origin of the new Omicron variant. According to those details, the Netherlands’ RIVM health institute found omicron in…

Days before an advisory panel to the Food and Drug Administration is expected to meet to discuss the Merck COVID-19 pill, the company revised the results of a previous study to reflect the pill is less effective than previously thought. The original Merck study, which was released early last month, found Molnupiravir reduced hospitalizations and…

Both the Centers for Disease Control and the Food and Drug Administration approved booster shots from Pfizer and Moderna for all adults Friday. Before Friday, only the Johnson & Johnson booster shot was approved for all adults. The Moderna and Pfizer booster shots had only been approved for seniors, adults who live in long-term care…

Just two days after Pfizer inked a deal with the United Nations’ Medicines Patent Pool (MPP) to allow generic drug companies to produce the company’s COVID-19 pill for use in 95 countries, the company announced Thursday it has made another deal. This time, it’s with the U.S. government. The COVID-19 pill deal is worth $5.29…
Welcome back to trustworthy journalism.
Here at Straight Arrow News, we…
Report without partisan spin, earning distinctions from media bias experts at AllSides and Ad Fontes
Share what other news outlets are underreporting with our Media Miss™ tool
Provide perspective with opinions and commentary from all sides





